Advancing a Key Artificial Pancreas Technology
Stable liquid glucagon developed by JDRF partner Xeris evaluated in first clinical study
The JDRF Artificial Pancreas (AP) project is focused on accelerating the development of commercially-viable AP systems that better mimic the biological function of the pancreas and reduce the daily burden of managing type 1 diabetes (T1D). To achieve this goal, JDRF funds programs across a strategic AP development plan. Each step in the plan represents incremental advances in AP system automation beginning with basic devices that shut off insulin delivery to prevent episodes of low blood sugar and progressing to fully automated, multi-hormone systems that maintain blood glucose at a target level. Basic AP systems are now reaching the market, but the more complex, fully automated systems require technologies that don’t exist today – like multi-hormone pumps, faster acting insulin, and stable liquid glucagon – so JDRF is helping create them.
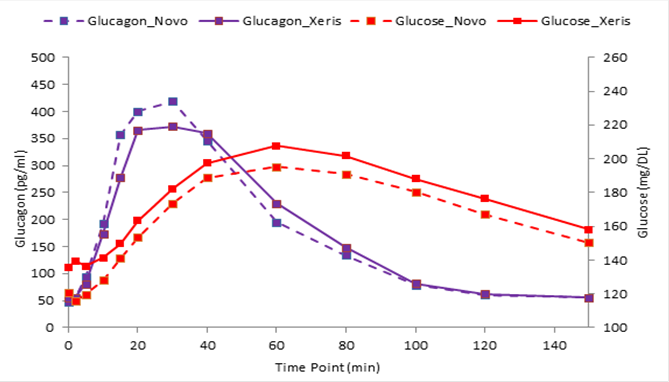
Last year, JDRF partnered with Xeris Pharmaceuticals to solve one of these technical hurdles –development of a stable, liquid glucagon compatible with T1D pumps. At a recent meeting of the Diabetes Technology Society, Xeris presented the results of the first clinical study evaluating their novel glucagon product; they call G-Pump™, in people with T1D. The goal of the study was to assess the safety, speed of absorption, and onset of action of G-Pump compared to an emergency rescue glucagon product marketed by Novo Nordisk under the brand name GlucaGen®, both delivered via an OmniPod® pump. On treatment day one, nineteen people with T1D received either the Xeris or Novo Nordisk glucagon via a pump and had their blood levels of glucagon and glucose measured for several hours. Then, on a subsequent day, each person returned for treatment day two, and received the other (Xeris or Novo Nordisk) glucagon, in order to compare the two types of glucagon in each person.
G-Pump glucagon effectively increased blood glucose levels in proportion to the dose given. G-Pump glucagon also produced blood glucagon levels similar to those of freshly prepared GlucaGen. Overall the results strongly support moving G-Pump glucagon to the next phase of development – clinical testing as a component of a multi-hormone AP system, which could move us one step closer to the development of more advanced AP systems.
For more information or to support JDRF’s artificial pancreas research program, please click here.