JDRF Partners with Thermalin on Ultra-Rapid Insulin
A “fast-on” and “fast-off” ultra-rapid insulin formulation could improve post-meal glucose control and enable advanced-generation artificial pancreas systems.
In an expansion of its fast-acting insulin research program, JDRF recently formed a partnership with Thermalin Diabetes to develop a new ultra-rapid-acting insulin formulation to help better manage type 1 diabetes (T1D). The partnership is built on a multi-year plan to develop novel types of insulin that begin working very fast and stop working very fast, more closely mimicking the action of natural insulin (non-diabetes). Such a product would be a significant improvement over currently available rapid-acting insulins that continue to act for hours after an infusion or injection, often leading to overdosing of insulin and potentially dangerous low blood-glucose levels, especially long after meals. A novel ultra-rapid-acting insulin could allow users to administer an insulin dose closer to mealtimes with less worry about delayed post-meal low blood-sugar levels. It would also be a key component of future advanced-generation artificial pancreas systems, enabling more precise insulin dosing and resulting in improved glucose control, especially around mealtimes.
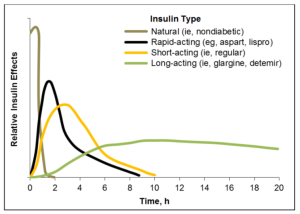
Studies comparing currently available rapid-acting insulins to natural or non-diabetes insulin show the clear differences in their action over time following use. Natural insulin begins working very rapidly in a person without T1D and, perhaps more importantly, stops working very quickly too. No currently available insulin product comes close to matching this pattern of action. The most noticeable difference is how much longer insulin products continue working after an injection compared to natural insulin. The slower onset of action and the extended duration of action make it especially hard to dose these products around mealtime, greatly complicate the dosing calculations built into artificial pancreas systems, and are a primary cause of post-meal low glucose events.
Founded in 2009, Thermalin Diabetes is pursuing novel insulin products to improve insulin therapy for people with diabetes and to enable new delivery technologies. The company is privately held and has previously been funded through a combination of private investments and grant funding from the National Institutes of Health and other sources.