FDA Approved! Abbott FreeStyle Libre® 3 Continuous Glucose Monitor, Ages 4+
It’s worth celebrating every time the FDA approves a device that makes is easier for people with type 1 diabetes (T1D) to manage their disease. Today’s announcement fits that bill. The FDA cleared the Abbott FreeStyle Libre 3 continuous glucose monitor (CGM) for children and adults ages 4 and up with diabetes, giving people additional choice for devices.
Another Tool in the Toolbox
CGM technology has revolutionized the treatment and management of T1D. Ever since the landmark 2008 study, funded by JDRF, that showed the benefits of CGM, their adoption has significantly increased in type 1 and is also increasing in type 2 diabetes. And, in that time, the devices have become increasingly accurate, easier to insert, more convenient to use, and covered by payers.
Tools like this will help people with T1D do better until cures are realized.
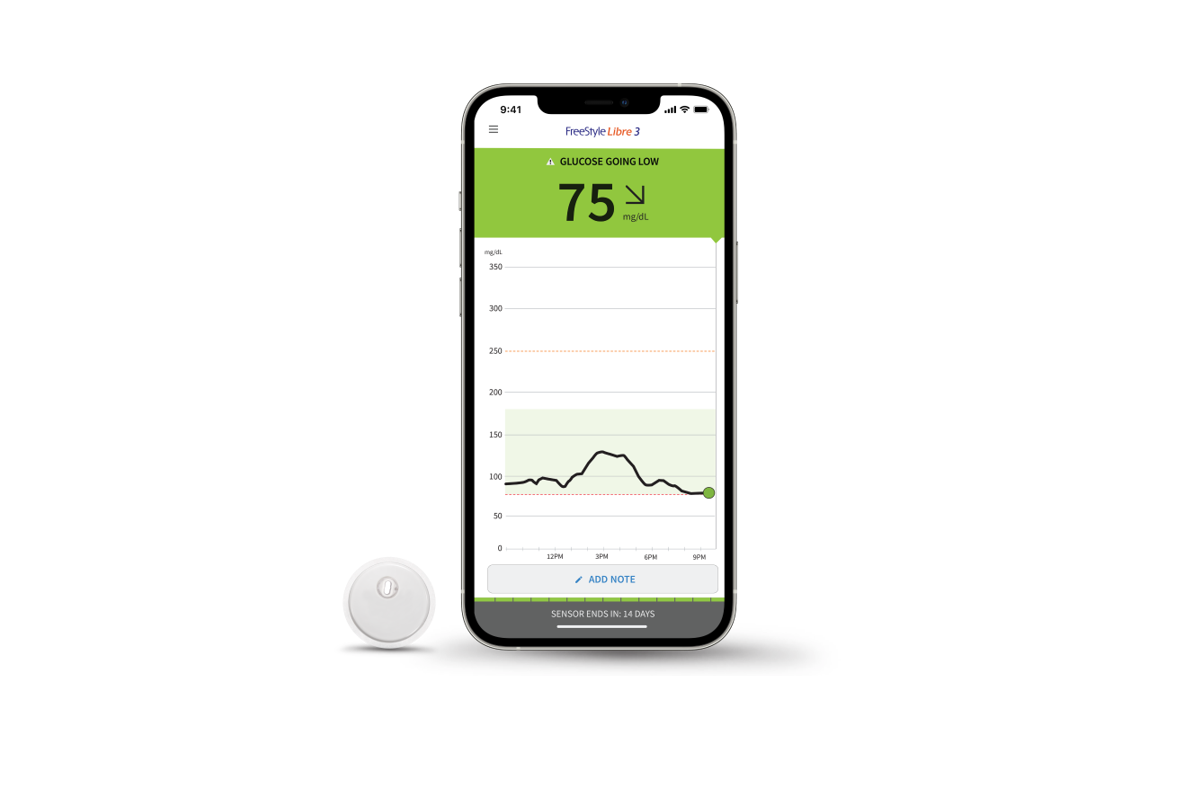
Specs
FreeStyle Libre 3 is an evolution of the FreeStyle Libre 2. It has many of the same features as its predecessor; it is approved for use on the arm, transmits data every minute to a smartphone via an app for both iOS and Android; and it features several notable improvements. This includes:
- A 70% smaller sensor. Per Abbott, this device is the size of two pennies stacked on top of each other.
- A Mean Absolute Relative Difference (MARD) of 7.9%. MARD is a key metric used to evaluate CGM accuracy by comparing CGM values to an established blood glucose test. Per Abbott, the Freestyle Libre’s MARD of 7.9% makes it the first CGM to have a MARD below 8%.
- Bluetooth range of up to 33 feet.
“JDRF is thrilled that the Abbott FreeStyle Libre 3 received clearance from the FDA,” says JDRF Associate Director of Research, Jonathan Rosen, Ph.D. “Rigorous research has shown many times over that CGM devices improve glucose control and other health outcomes. A major priority for JDRF is for people with type 1 diabetes to have access to and choice of effective, convenient technology for glucose management, and this is a step forward in that direction.”
JDRF—A Key Driver of CGM
JDRF has played a pivotal role in novel CGM development, as well as access and adoption, including supporting a clinical trial that conclusively demonstrated that CGM use improved health outcomes for people with diabetes. Once it becomes available in the coming months, FreeStyle Libre 3 will be one of several commercially available CGM systems, giving people with T1D the freedom to choose the tools and systems that are right for them.
JDRF will continue to monitor the field and push for meaningful technological advancements, and we won’t ever stop fighting for affordability, choice, and coverage on our path to a world without T1D.
Additionally, FreeStyle Libre 3 is the CGM component of an automated insulin delivery (AID) system, also referred to as artificial pancreas system, currently being developed in Europe by CamDiab and Ypsomed. JDRF funded Roman Hovorka, Ph.D., of the University of Cambridge and Director of CamDiab, from 2006-2019 to develop the algorithm, which is approved in Europe and UK, in that system.